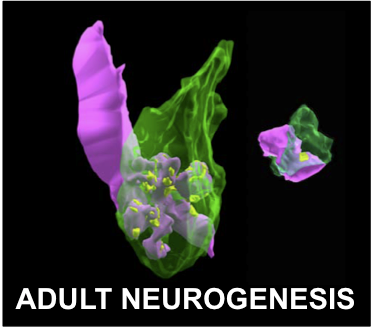
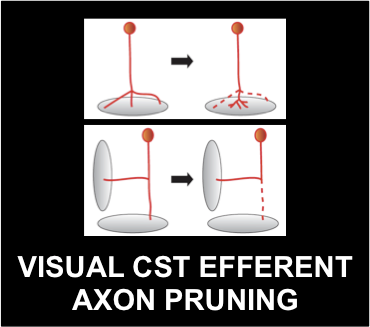
In the developing central nervous system, neuronal precursors undergo a series of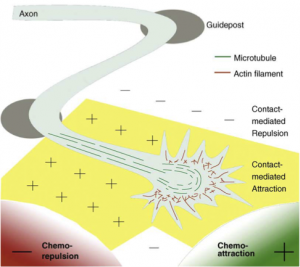
progressive events, including cell division, cell differentiation, cell migration, and neurite extension, to initially establish an excess of connections. These neurons subsequently undergo a series of regressive events, including cell death, synapse elimination and neurite pruning to refine the connections. Through these intricate developmental processes, extensive neural networks emerge that are functional and highly complex. Abnormalities in these processes are known to cause neuropsychiatric disorders including autism and schizophrenia.
The Cheng Laboratory uses a diverse range of model organisms such as the C. elegans, mouse, and ferret to comparatively study how axonal connections are established and refined throughout development. We use integrative approaches to gain a comprehensive understanding of axon refinement at the molecular, cellular and systems level.
We specifically investigate the following questions:
- What molecular signaling pathways and neural activity mechanisms are involved in proper axon guidance and axon refinement in the developing brain?
- How do molecular signaling pathways and neural activity mechanisms interact to influence axon guidance and axon refinement in the developing brain?
- Are the molecular signaling pathways and neural activity mechanisms determining connectivity of newborn neurons in the young developing brain different in the old aging brain?
- In neuropsychiatric disease models, can we rescue defects by targeting the molecular signaling pathways and neural activity mechanisms discovered to be involved in axon guidance and axon refinement?
Our current projects study the following: